Overview
Woodland creation and management deliver a wide range of environmental benefits. The extent of those benefits is determined by a range of factors, including the type of woodland, the way it is managed, and its position in the landscape.
This Research Note summarises current evidence on how the management, creation, and configuration of woodlands within a landscape affect biodiversity, ecosystem resilience, and key environmental benefits.
The Research Note concludes by discussing the potential synergies and trade-offs between different environmental benefits, and their interaction with social and economic outputs.
Introduction
The UK is committed to the protection, expansion, and sustainable management of woodlands to secure a wide range of environmental and social benefits. Well-managed woodlands of any type can enhance biodiversity and support many protected species.
Additionally, woodlands provide important benefits to people through regulating and provisioning ecosystem services such as timber and wood products, climate change mitigation, water quality improvements, and recreation. However, many woodlands have fallen into unfavourable or intermediate ecological condition due to a lack of suitable management, or to external pressures such as herbivore damage, invasive species, and pests and diseases (National Forest Inventory, 2020).
The type, location, and management of woodland elements within a landscape, and the potential network they form, determine the extent to which they support native biodiversity and contribute to the delivery of ecosystem services. Lawton et al. (2010) identified four key attributes for managing, restoring, and creating habitats across a landscape to benefit biodiversity: better, bigger, more, and joined.
For woodland networks, these are implemented through the management of existing woodlands to improve their condition (better), and through woodland creation (more). New woodlands can be created as spatially discrete woodlands, which could provide stepping stones between nearby woodland fragments (joined), or adjacent to existing woodlands to expand them (bigger). The location of new woodland and its proximity to other wooded features, including trees outside woodlands, hedgerows, or wood pasture, may also influence connectivity across a landscape.
This Research Note summarises current evidence on how the management, creation, and spatial configuration of woodlands within a landscape affect biodiversity, ecosystem resilience, and key environmental benefits including climate change mitigation and adaptation, flood mitigation, and improved water quality. We primarily focus on the benefits to woodland-associated biodiversity, while also highlighting where actions can deliver benefits to other species.
The roles of trees outside woodlands and other wooded habitats, such as wood pasture, are not specifically covered in this review.
The evidence has been gathered from technical reports, guidance documents, and the scientific literature, including review articles and meta-analyses. We present the findings in two sections: ‘Managing existing woodland’ and ‘Creating new woodland’, each with a table of summary statements that describes the strength and agreement of the evidence (IPBES, 2018).
In the final section, we consider trade-offs between biodiversity and productive forestry, as well as between different environmental outcomes. We also highlight some of the potential synergies in woodland management and creation to achieve multiple environmental benefits, including those beyond the scope of this Research Note.
Managing existing woodland
Woodland management is the maintenance, improvement, and utilisation of existing woodlands through forestry operations and land management activities. Common management objectives include biodiversity enhancement, carbon sequestration, and the production of timber and wood products.
There are likely to be synergies and trade-offs between different management objectives depending on factors such as woodland type and operational approach (Sing et al., 2018). These are discussed in more detail in ‘Synergies and trade-offs’.
Managing existing woodlands to improve their ecological condition has been demonstrated to be a highly effective method of enhancing woodland-associated biodiversity and ecosystem service delivery across a landscape, particularly when ancient woodlands and neighbouring habitats are targeted (Crick et al., 2020).
Here we review the evidence supporting the improvement of woodland ecological condition by increasing structural complexity, increasing tree and shrub species diversity, and reducing external pressures.
Increasing structural complexity
Structural complexity describes the variation in woodland structure and is influenced by the woodland’s underlying topography, hydrology, and geology. Vertical structural complexity (Figure 1) captures the height and age ranges of tree and understory species and includes features such as deadwood, veteran trees, and patches of scrub.
The variation in stem density and diameter across a woodland, and the distribution of open areas, bodies of water, and rides, is defined as horizontal structural complexity.
Benefits for wildlife
High structural complexity is correlated with a higher diversity of microclimates and ecological niches that benefit a greater number of species.
Research has demonstrated the positive impact of structural complexity on the abundance and species richness (Box 1) of birds, bats, small mammals, flying insects (Fuller et al., 2018), and vascular plants (Humphrey et al., 2015; Waddell et al., 2024). Certain features have been shown to benefit specific groups, such as areas of lower tree density which are favoured by bats (Fuentes-Montemayor et al., 2013).
Early woodland developmental stages are equally important as they support unique species assemblages, particularly among woodland birds (Wilson et al., 2006; Sweeney et al., 2010).
Figure 1 – Sustainable woodland management to improve biodiversity value and provide wider environmental benefits.
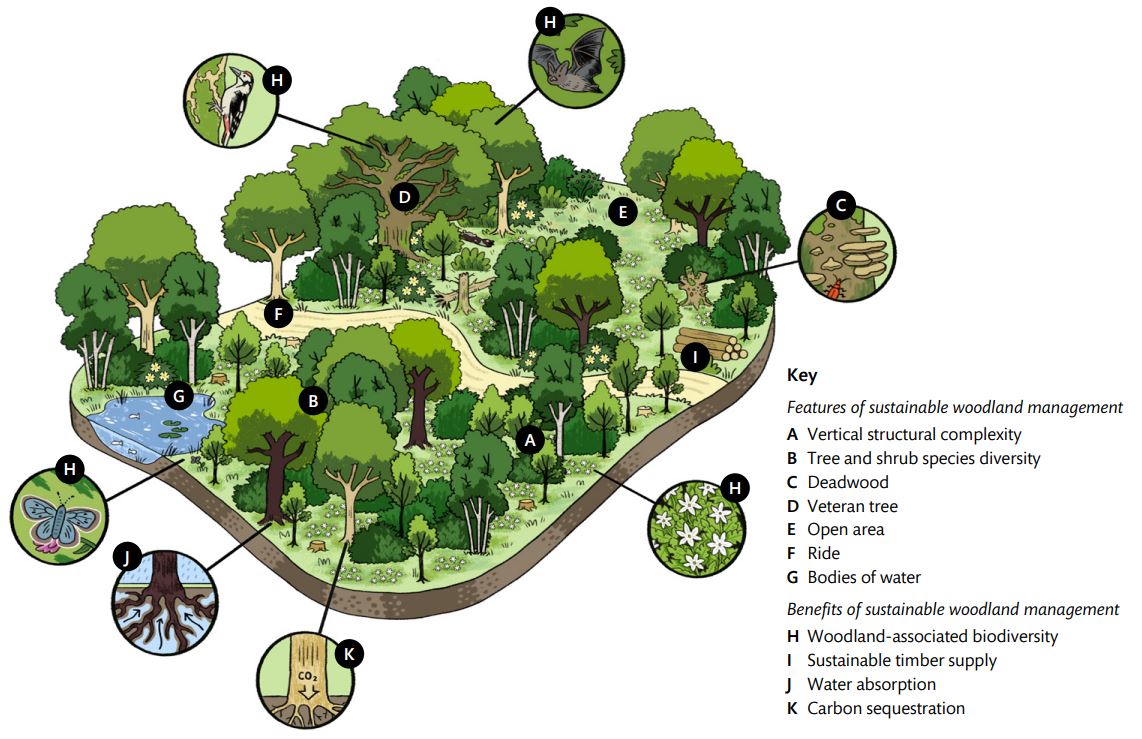
Note: Broadleaved woodland is represented, but example features and benefits, also apply to mixed and coniferous woodlands. Variation in the distribution of structural features across a woodland is referred to as horizontal structural complexity.
Box 1 – Abundance and species richness
These ecological terms are commonly used to summarise the amount of biodiversity of a given taxonomic or functional group within an area. ‘Abundance’ refers to the total number of individuals of that group, while ‘species richness’ describes the total number of species. The two are not always correlated and it is possible to have abundant woodlands with low species richness and habitat that is highly species rich but at a low abundance.
Ancient and veteran trees add further complexity and are important for the abundance and diversity of many forest-associated species (Zeller et al., 2023). Deadwood (fallen, standing, or as part of a living tree) is a component of healthy woodland ecosystems and provides a suite of unique ecological niches.
The total volume, species, and decay stage of deadwood determine which associated species are present, and studies have positively correlated the species richness of bryophytes, fungi, lichens, and invertebrates with one or more of these factors (Lassauce et al., 2011; Andringa et al., 2019; Tomao et al., 2020).
Canopy gaps are important features that influence the biodiversity supported by mature woodlands. They allow light to penetrate the woodland interior, potentially to the forest floor, which benefits a range of species, particularly ground vegetation, invertebrates, and lichen (Zeller et al., 2023).
Glades, rides, and clearings provide larger patches of open and edge habitats that can also benefit different species assemblages and contribute to overall biodiversity (Iremonger et al., 2006).
The decline in traditional management practices, such as coppicing, in favour of uniform and closed-canopy timber plantations has led to a significant decline in forest biodiversity across Europe, particularly affecting species that thrive in more open environments (Kirby, Buckley, and Mills, 2017).
Structural complexity may also bring resilience to a woodland. There is evidence that reduced tree density can increase resilience to pests and pathogens (Roberts et al., 2020), while a greater variety of micro-habitats may mitigate climate change impacts on resident biodiversity (Box 2) (Bellamy et al., 2018).
Box 2 – Woodland resilience and adaptation
In the context of woodland ecosystems, ‘resilience’ and ‘adaptation’ are interlinked but distinct terms which describe how a woodland may react to external pressures and a changing climate.
‘Resilience’ describes the capability of a woodland to withstand or recover from an external pressure or shock while maintaining the core species and features that allow it to host biodiversity and deliver ecosystem services (Bellamy et al., 2018). Common pressures include pests, diseases, invasive species, and environmental events such as droughts and storms which bring high winds and heavy rainfall. Many of these threats are becoming more frequent and severe due to climate change.
‘Adaptation’, on the other hand, is the capacity of the woodland to alter in response to changing environmental conditions such that it can continue to host biodiversity and provision ecosystem services into the future. Adaptive potential can be introduced to a woodland through changes at different levels, such as in species composition by planting non- or near-natives, or in the genetics of the tree species by sourcing seed from southerly provenances (i.e. assisted migration) or including individuals selected for desirable traits (such higher drought tolerance).
Other environmental benefits
Woodlands with higher structural complexity have been shown to accumulate more above-ground carbon1K than less structurally complex forests due to complementary resource use by trees of different sizes (Ali, 2019). Retaining natural deadwood and brash from forestry operations increases the amount of carbon stored in forest soils (James et al., 2021).
Fallen deadwood also adds roughness to the woodland floor which contributes to flood mitigation and better water quality by increasing water infiltration and absorption1J, slowing water run-off, and reducing soil loss (Nisbet et al., 2011).
Increasing tree and shrub species diversity
Closely associated with structural complexity is the variety and spatial distribution of tree and shrub species in the canopy and understory1B. The functional traits of each tree and shrub species (e.g. the level of shade cast, the water-holding capacity of its bark, and the nutritional value of its leaf litter) affect the community of associated species and the delivery of ecosystem services (Mitchell et al., 2021).
There is a current focus on increasing the proportion and diversity of broadleaved species to support native ecological communities. This approach is beneficial for various woodland types and frequently takes precedence in restoration efforts of non-native woodlands, such as Plantations on Ancient Woodland Sites (PAWS) (Box 3).
Benefits for wildlife
A high diversity of tree species is well understood to be a key driver of woodland-associated biodiversity, but notably when species with different functional traits are combined, rather than when there is simply a higher number of tree species (Ampoorter et al., 2020).
In many mixed and coniferous woodlands, increasing the proportion of broadleaves is likely to have positive biodiversity outcomes for many different taxa, particularly where native tree species such as oak are incorporated (Broome, Stokes, et al., 2021; Zeller et al., 2023). However, an increased number of broadleaved tree species may not be appropriate in certain situations, such as in native pinewoods or red squirrel strongholds (Forestry Commission, 2012).
Other environmental benefits
Many woodlands of multiple species have been shown to sequester more above-ground carbon than monocultures due to complementarity between species and overyielding (Williams et al., 2017; Warner et al., 2023). Mixed stands can have greater resilience to external pressures and disturbances such as specialist insect pests, fungal pathogens, and weather events predicted to intensify under climate change, including drought and storms (Jactel et al., 2017; Messier et al., 2022). The strength of these effects, however, can depend on both the tree species in the mixture and wider site factors.
The effect on ecosystem service delivery of increasing the proportion of broadleaves in mixed woodlands is likely to be highly context dependent. Certain broadleaved species are better adapted to riparian and floodplain contexts, where they can capture sediment and slow flows while providing additional ecological benefits (Nisbet et al., 2011; Burton et al., 2018). On upland catchment slopes, coniferous species may intercept more rainfall and promote higher infiltration than most broadleaved species, thereby slowing surface run-off (Cooper et al., 2021).
Surface water acidification has negative ecological and environmental consequences. The effect of tree type on water acidification is complex and may be less important than woodland location and maturity (Nisbet and Evans, 2014). In the acid-sensitive uplands, conifers are generally more acidifying than broadleaves as they are more effective at scavenging atmospheric acid pollutants (Nisbet et al., 2011). On the other hand, alder, a native broadleaved species, can cause local acidification in poorly buffered soils due to nitrate leaching following nitrogen fixation by root nodules (Nisbet and Evans, 2014).
Box 3 – Woodland
Many UK woodlands are in unfavourable ecological condition and require restoration to support biodiversity recovery and establish a resilient ecological network.
Restoration approaches primarily focus on increasing native tree cover, safeguarding remnant ecological features, and addressing external pressures. Non-native woodlands, including Plantations on Ancient Woodland Sites (PAWS), are often targeted for these actions.
PAWS are areas of ancient semi-natural woodland that have been cleared and replanted with non-native trees. These sites span a continuum ranging from monocultures of non-native conifers to a mix of native and non-native species. They can negatively impact wildlife reliant on native trees (e.g. pied flycatcher and purple emperor butterfly), and can harm native flora through competition from non-native species. The evidence base for restoration is still emerging, and environmental benefits will vary depending on site-specific factors and the approach adopted.
Removing non-native conifers and reinstating native broadleaves can allow the re-establishment of native ground flora (Kirby, Goldberg, and Orchard, 2017) and create more available niches for wildlife (Quine et al., 2007). A gradual removal strategy has been shown to avoid unintended negative impacts on shade-adapted specialists (Brown, Curtis, and Adams, 2015; Broome, Inchboard, et al., 2021) or species that utilise conifer habitats (Trout et al., 2012).
The effect of tree type on carbon sequestration and greenhouse gas mitigation is also highly nuanced and dependent on species, site, management, and the substitution impacts of any harvested wood products (Körner, 2017). Over short timeframes (e.g. up to 50 years), fast-growing productive conifers generally sequester and substitute the greatest amount of carbon (Forster et al., 2021; Matthews et al., 2022).
However, after longer periods of time, the carbon sequestration between different woodland types becomes more equal as long-lived broadleaved species accumulate large above- and below-ground carbon stocks (Mayer et al., 2020; Matthews et al., 2022).
Reducing pressures
There is a strong evidence base supporting our understanding of the causes and impacts of woodland degradation by biotic and abiotic pressures. For example, the National Forest Inventory Woodland Ecological Condition demonstrates that pressures such as pests and diseases, invasive non-native plants, and damage by wild and domestic animals are major factors contributing to unfavourable woodland ecological condition in Great Britain (National Forest Inventory, 2020).
Here we summarise the evidence for local-scale management interventions to reduce pressures in woodlands. Some pressures, such as pollution damage, require broader management or policy actions beyond the woodland area and are not covered here. Evidence for the effectiveness of specific measures is still emerging as there is often a delay between action and biodiversity response (Watts et al., 2020).
Removing or reducing negative impacts will likely benefit woodland biodiversity and support the recovery of many threatened or priority species, particularly woodland specialists (Alder, Fuller, and Marsden, 2018).
Benefits for wildlife
The negative impacts of over-browsing by herbivores, such as deer, on understory vegetation are well documented (Kirby, 2001; Ramirez, Jansen, and Poorter, 2018), and these changes have been reported to adversely affect woodland birds (Fuller et al., 2007), small mammals (Flowerdew and Ellwood, 2001), and invertebrates (Stewart, 2001). One study found that exclosure of deer from coppiced woodlands resulted in higher density and cover of understory vegetation, as well as a higher abundance of songbirds2D, after three years of coppice growth (Gill and Fuller, 2007).
Similarly, invasive plants (e.g. rhododendron) alter the structure and composition of woodlands, which will in turn alter the availability and distribution of resources, influence animal behaviour, and affect animal-plant interactions (Manchester and Bullock, 2000; Vilà et al., 2011; Stewart et al., 2021). Tree diseases, such as ash dieback, can cause rapid mortality in the affected species with potentially severe negative impacts on populations of associated species and the provision of ecosystem services (Mitchell et al., 2014).
Climate change will present substantial challenges to woodlands due to shifts in temperature, altered rainfall patterns, and extreme weather events (Ray, Morison, and Broadmeadow, 2010; Patacca et al., 2023). Rising average temperatures may also increase the spread of pathogens and diseases (Frederickson-Matika and Riddell, 2021; Inward, 2023). Addressing other external pressures and maintaining functionally varied tree stands within well-connected woodlands can improve woodland resilience and facilitate species adaptation to climate change (Ray, Morison, and Broadmeadow, 2010).
However, empirical evidence supporting effective wildlife adaptation measures in woodlands remains limited.
Figure 2 – Example external pressures on woodlands which, when reduced, can enhance the woodlands’ wildlife value and the delivery of environmental benefits.
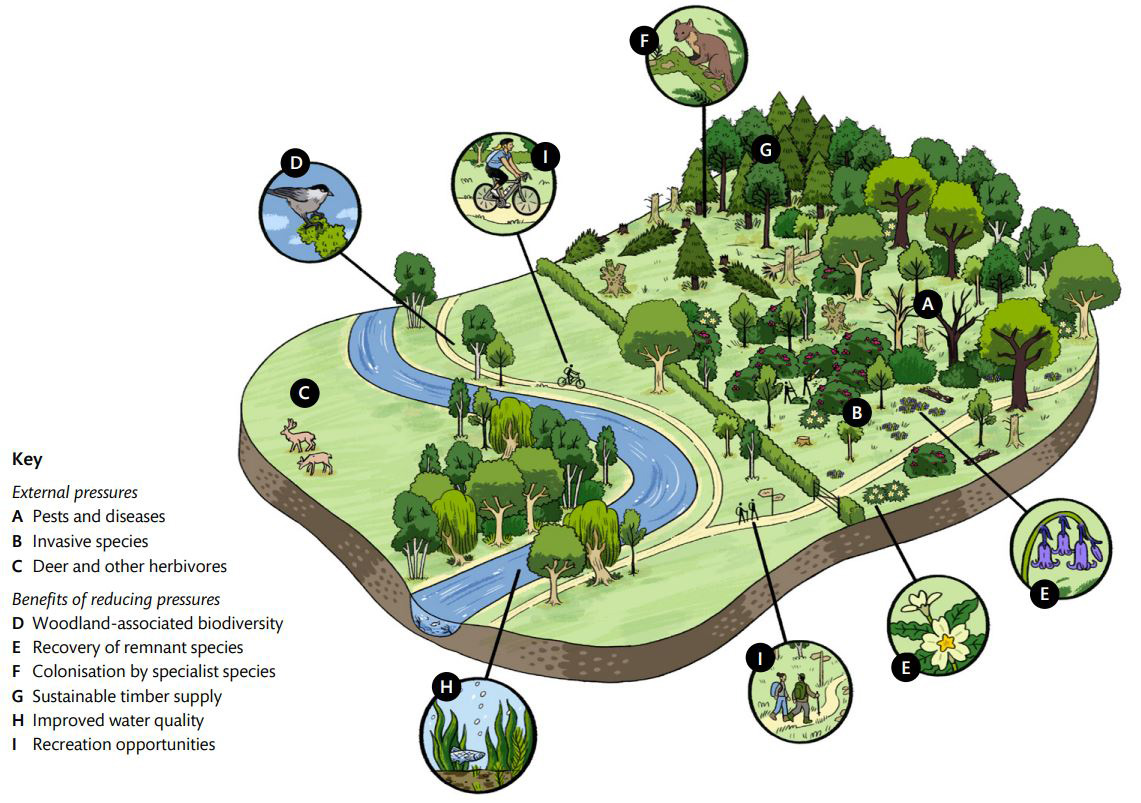
Note: Example pressures and potential benefits of effective management are annotated and referenced with superscript letters in the text. Climate change represents an additional pressure to woodlands that will likely exacerbate the threats from pests and diseases (A) and invasive species (B).
Other environmental benefits
Removing or reducing pressures on woodlands will protect biodiversity and enhance ecosystem functions and processes that deliver wider environmental benefits. For example, enabling understory regeneration through deer control can increase the above-ground carbon stored in woodlands (Tanentzap and Coomes, 2012). Allowing regeneration of ground flora may also improve water regulation and flood mitigation services provided by woodlands, although this has not been tested directly.
Creating new woodland
Woodland creation is the establishment of new woodland on land not previously wooded. New areas of woodland provide habitat, deliver important environmental benefits, and complement existing mature woodland, hedgerows, wood pasture, and lone trees to create diverse wooded landscapes.
Increasing woodland cover in a landscape is well known to benefit many taxa, including birds, mammals, fungi, vascular plants, and certain invertebrate groups (Humphrey et al., 2013; Bowler et al., 2023). While it may take years before these benefits are fully realised (Fuentes-Montemayor et al., 2022), the developmental stages following creation will benefit different species assemblages before a mature woodland is achieved.
Woodland can be established as new spatially discrete woodlands, which can act as stepping stones to improve landscape connectivity, or as an expansion of an existing woodland, which can be colonised more rapidly. Here we review the benefits of woodland creation by these two approaches.
Spatially discrete woodlands
The composition and structure of new spatially discrete woodlands, and their proximity to other wooded sites and features, will strongly influence their success as habitats and their capacity to deliver ecosystem services.
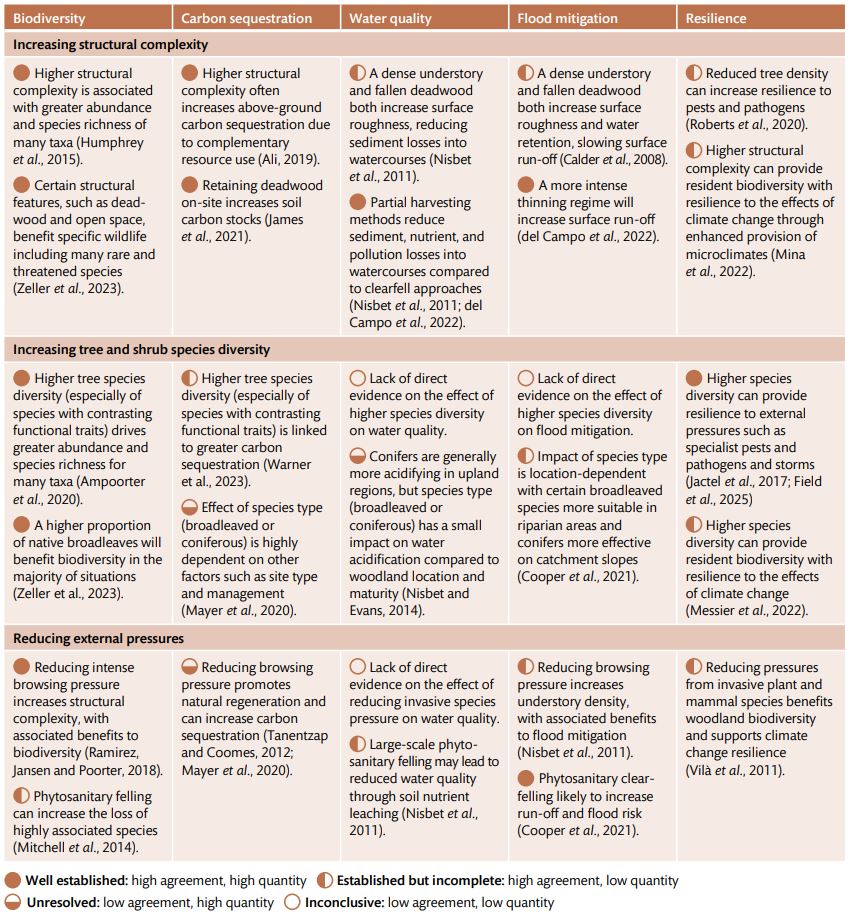
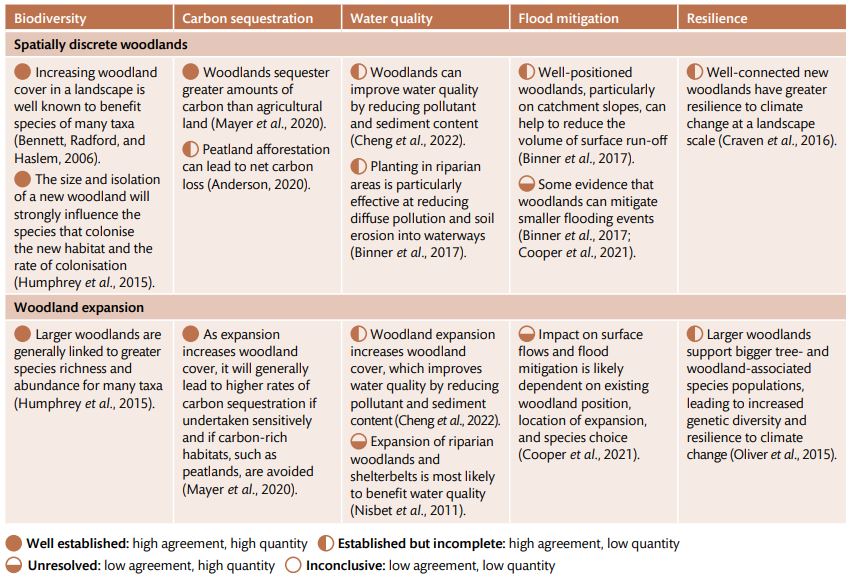
Table 1 Summary of the evidence base (quantity and agreement of evidence) on the biodiversity outcomes and environmental benefits of woodland management actions aimed at improving woodland condition.
Benefits for wildlife
The size of a new woodland is an important spatial consideration. Bigger woodlands tend to support both a higher number of species and larger population sizes of individual species. This is due to both the larger area of continuous woodland and the increased likelihood of a greater abundance and diversity of micro-habitats to provide shelter and resources.
Larger population sizes in bigger woodlands are typically reflected by higher levels of genetic diversity, which can allow populations to adapt more readily to changing conditions (Bellamy et al., 2018). Many taxa, including birds (Whytock et al., 2018), vascular plants (Waddell et al., 2024), and certain bryophyte and invertebrate species are more abundant and have higher species richness in larger woodlands (Humphrey et al., 2015). For other taxa, such as many small mammal species and some invertebrates, the significance of woodland area is conditional on other factors, such as habitat quality or isolation (Humphrey et al., 2015; Fuentes-Montemayor et al., 2020).
Woodland isolation is a product of its geographic position, its proximity to other trees and woodlands, and how easily species can move through the surrounding landscape. The degree of isolation of a newly established woodland, and the age of the nearby existing forests, will influence the species that a new woodland can host and the rate at which those species colonise, with different taxa and functional groups responding differently (Humphrey et al., 2015).
For slow-dispersing taxa, such as many woodland specialist vascular plants and fungi, proximity to established woodland is a major factor determining colonisation success (Scheidegger and Werth, 2009). Proximity to other wooded habitats, such as wood pasture, parkland, and areas of scrub, may also increase connectivity from existing woodland habitat to sites of afforestation (Prevedello and Vieira, 2010).
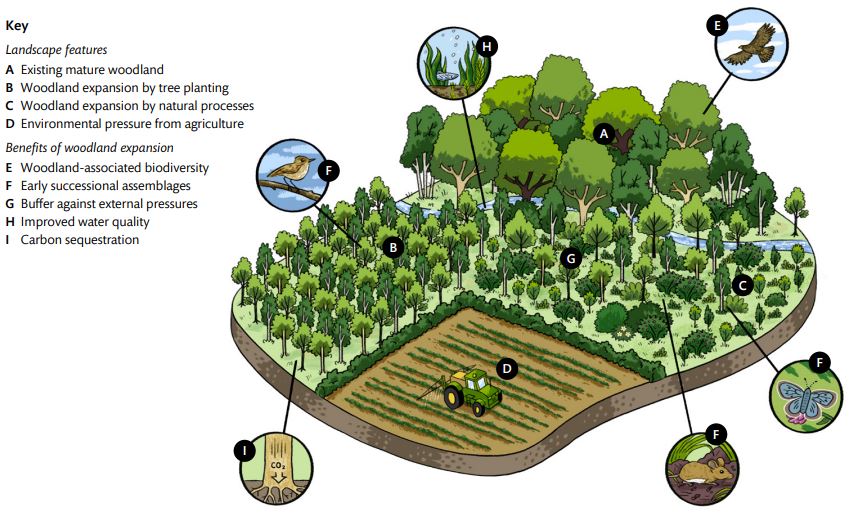
Figure 3 – Landscape features and benefits of spatially discrete woodland creation.
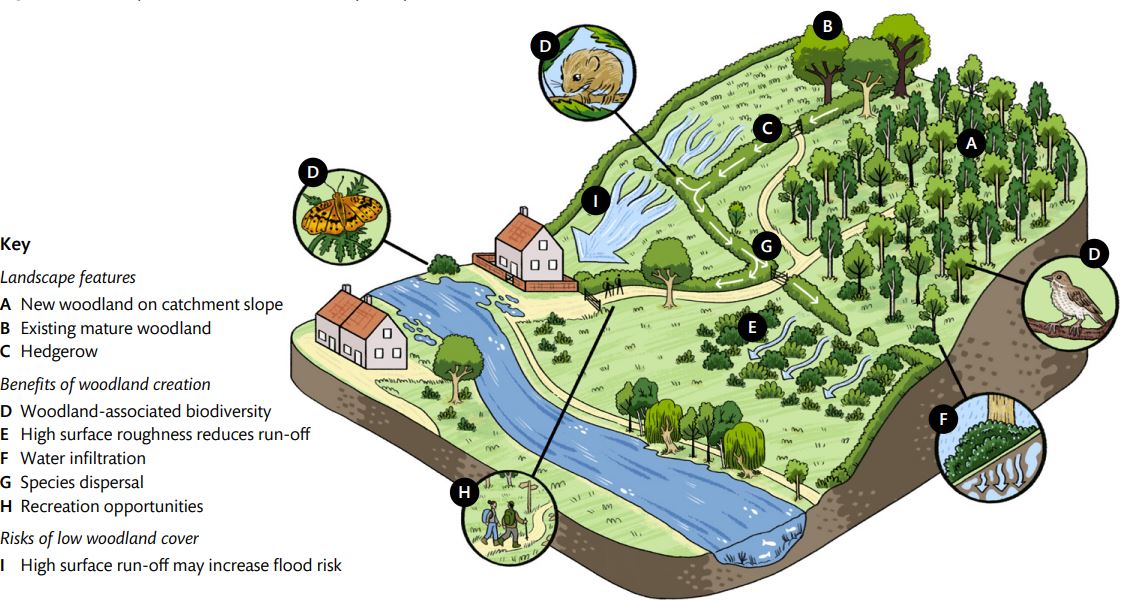
Note: Example landscape features, potential benefits of woodland creation, and risks associated with low woodland cover are annotated and referenced with superscript letters in the text. The positioning of new woodland creation (A) within the topography of a catchment will affect the delivery of environmental benefits such as flood mitigation (E and F). Isolation of a new woodland, relative to other wooded features, will impact the rate of colonisation by local biodiversity (G).
Other environmental benefits
Woodland soils are typically undisturbed and well-structured compared with those of other open land uses. This leads to greater water infiltration which improves water storage and reduces surface run-off, soil nutrient leaching, and topsoil erosion during heavy rainfall (Nisbet, 2020; Cheng et al., 2022).
The total amount of new woodland cover, its development through time, and its distribution within a catchment will impact the effectiveness of these new wooded areas for both flood mitigation and water quality improvement. When established on the slopes of catchments, particularly in a cross-slope configuration, woodland soils can absorb surface water flowing neighbouring agricultural land, slowing run-off and preventing topsoil erosion (Cooper et al., 2021). However, afforestation in drought-prone areas may have negative effects on water yield during dry weather (Buechel, Slater, and Dadson, 2022).
Establishing new woodlands in a river floodplain can lessen peak flood magnitude, particularly for smaller flood events, and modelling studies suggest floodplain afforestation could decrease the flow rate, raise the water level, and delay peak discharges (Dixon et al., 2016; Cooper et al., 2021).
Riparian woodlands may also contribute to reducing peak flood flows and have been shown to improve water quality by intercepting pesticide drift and reducing sediment run-off (Binner et al., 2017). By shading watercourses, they also lessen thermal stress to fish and subdue weed growth (Nisbet et al., 2011). While it is recognised that higher woodland cover can provide greater mitigation benefits for small-scale floods, woodlands do not appear to provide any significant mitigation for extreme flooding events (Nisbet et al., 2011).
Generally, woodlands sequester more atmospheric carbon than agricultural land uses and are significant long-term carbon sinks (Morison et al., 2012). Creating new woodlands is therefore likely to greatly increase landscape-scale carbon sequestration (despite potential initial carbon losses from soil disturbance) if undertaken sensitively, and avoids carbon-rich habitats that do not habitually support tree cover (Mayer et al., 2020; Seddon et al., 2021). Peat-forming wet woodlands have the potential to make disproportionately high contributions to carbon sequestration despite generally small woodland areas (Milner et al., 2024).
Larger woodlands will sequester greater amounts of carbon, as more land area will become wooded. Forest edges may accumulate more above-ground biomass than woodland interiors due to adjacent nitrogen deposition, suggesting that woodland area spread across multiple smaller sites may store more above-ground carbon (Meeussen et al., 2021; Morreale et al., 2021). The location of afforestation activities may also impact net carbon sequestration through both the land cover type it replaces and the growth rate of the trees established at that site.
Woodland expansion
Woodland expansion is the enlargement of an existing site by adjacent woodland creation. Compared to spatially discrete woodland creation, woodland expansion affords a range of advantages to both the mature and newly established woodlands. Even greater benefits can be achieved if expansion is used to link multiple mature woodlands.
Expansion can be achieved through two methods: tree planting and natural processes (Box 4). Tree planting provides control over species composition and spatial distribution but can lead to greater ground disturbance and may bring pests and diseases onto the site.
Employing natural processes removes the biosecurity concern and may be less intensive and costly, but the outcome is unpredictable and species composition and positioning can only be controlled through enrichment planting and silvicultural management. Fencing or deer control may be required to protect self-seeded saplings if herbivore pressure is high.
Benefits for wildlife
Increasing the size of a woodland by tree planting or natural processes is well understood to boost its value for biodiversity in several ways.
Firstly, expansion leads to a bigger woodland which, as described above, can support higher species diversity and abundance. Secondly, expansion inherently develops horizontal structural complexity by creating new younger areas of woodland next to mature trees which will be inhabited by different species assemblages including, over time, woodland specialist assemblages. Thirdly, compared to isolated woodland creation, afforestation adjacent to existing mature woodland has been shown to grow more quickly and with higher structural complexity, further enhancing biodiversity (Hughes et al., 2023). Finally, the adjacency of expansion also speeds up colonisation from the mature woodland into the new habitat when compared to spatially discrete woodland creation, particularly for slow-dispersing species (Humphrey et al., 2015).
The method of expansion influences the species and communities that colonise the afforested area. The early successional scrub that is typically the result of natural colonisation in lowland areas provides a wildlife-rich and structurally diverse habitat that is favoured by small mammals, bird communities (Broughton et al., 2022), and invertebrates such as Lepidoptera (Merckx, 2015).
Tree planting is often supported by the suppression of such scrub, but generally delivers young woodland more quickly and with greater reliability. While this may benefit woodland-associated taxa, canopy closure limits light penetration and reduces biodiversity value until canopy gaps develop or are introduced through management.
Figure 4 – Landscape features and benefits of expansion of an existing mature woodland through tree planting and natural processes.
Box 4 – Natural colonisation and natural regeneration
Collectively termed ‘natural processes’, both ‘natural colonisation’ and ‘natural regeneration’ describe the growth of tree saplings from natural seed dispersal or vegetative reproduction. The difference between the terms is that natural regeneration occurs within a woodland or on land that was recently forested (such as following a clearcut or coup felling) whereas natural colonisation occurs on land that has not been woodland in recent history.
Other environmental benefits
Woodland expansion provides many of the same ecosystem services as spatially discrete woodland creation, but also presents its own opportunities and challenges. The higher growth rate of adjacent woodland creation may lead to greater carbon sequestration, while expanding a woodland through natural processes may avoid the initial carbon losses from the soil disturbance of tree planting. The method of expansion may also affect flood mitigation since young woodland is achieved more quickly through tree planting, but natural colonisation may result in a denser and more varied root network that enhances water infiltration (Harvey and Henshaw, 2023).
Enlarging a woodland also creates a buffer along the extended edge(s) that reduces negative edge effects and protects the mature site, its wildlife, and any internal water bodies from external environmental pressures, such as pesticide spray drift, pet disturbance, and extreme weather events (Crick et al., 2020).
Synergies and trade-offs
Woodlands are planted and managed to deliver multiple objectives. Understanding the potential synergies and trade-offs between these objectives will help planners and forest managers make informed decisions about the best approach to woodland management or creation.
Managing existing woodlands
Managing a woodland to maximise its biodiversity value often involves significant trade-offs with timber production and carbon sequestration (Sing et al., 2018).
Actions to improve woodland structure, such as establishing a diverse mixture of tree species or employing selective thinning regimes, can increase harvesting costs. The felling of premature plantation trees, such as in PAWS restoration, may be required to establish a native woodland community, but could cause the loss of a productive timber crop. Areas that are not designated as high value for biodiversity or other environmental benefits can be managed under an integrated approach to deliver multiple benefits while maintaining productivity (Sing et al., 2018).
Woodlands with high biodiversity value are known to also provide a range of other environmental benefits. Structurally complex and diverse riparian woodlands stabilise riverbanks and reduce sedimentation, thereby improving water quality and enhancing flood mitigation. The aesthetic, recreational, and cultural value of a woodland is also enhanced by high levels of biodiversity (Sing et al., 2018).
Recreation in woodlands is valued at around £907 million annually and provided health benefits to an estimated 3.2 million people in 2022, valued at £1149 million (Office for National Statistics, 2024).
Productive woodlands provide habitats for species and can deliver synergistic environmental benefits. For example, depending on their age, species, and location within the catchment, they can deliver flood mitigation benefits. Highly productive woodlands also sequester large amounts of carbon, although this may be off-set by carbon losses from harvesting.
Management operations provide access to woodlands (e.g. by creating roads and paths), although intensively managed sites may have less recreational value (Sing et al., 2018). The benefits of enhanced access will also need to be balanced against the potential for harmful impacts on biodiversity from increased woodland use (Marzano and Dandy, 2012).
Table 2 – Summary of the evidence base (quantity and agreement of evidence) on the biodiversity outcomes and environmental benefits of woodland creation actions.
Creating new woodlands
Woodland creation replaces an existing land cover, and the biodiversity value and ecosystem service provision that is lost may represent a significant trade-off. For example, establishing new woodlands on agricultural land could affect food production and security. However, there is limited evidence to fully assess the impact of woodland expansion on provisioning services such as food production in agricultural landscapes (Burton et al., 2018).
Woodland creation on existing non-woodland semi-natural habitats may increase carbon sequestration, but may also result in a net loss of biodiversity (Wilkes et al., 2020). Furthermore, potential environmental benefits may be greater from the creation or restoration of other semi-natural habitats compared to woodland in some locations. For example, in upland areas, heathland may have greater biodiversity and recreational value, while woodlands may provide greater carbon storage and timber (Cordingley et al., 2016).
The size of woodland creation sites may also present important trade-offs. Large woodlands have been shown to benefit many taxa (Humphrey et al., 2015) and to be buffered against external pressures. However, creating large woodland sites may not always be feasible due to constraints from other land uses. In these cases, focussing creation on expanding and joining existing smaller woodland fragments can be more beneficial than creating larger isolated woodlands (Synes et al., 2020).
Siting new woodlands close to urban areas can provide social, cultural, and health benefits by enhancing access to nature. Additionally, woodland creation near populated areas and close to pollution sources is likely to provide enhanced benefits from air pollution absorption (Powe and Willis, 2004). However, the woodland and biodiversity it hosts may be negatively affected by pollution and high footfall.
References
Alder, D.C., Fuller, R.J. and Marsden, S.J. (2018) ‘Implications of transformation to irregular silviculture for woodland birds: A stand wise comparison in an English broadleaf woodland’, Forest Ecology and Management, 422, pp. 69–78. Available at: https://doi.org/10.1016/j.foreco.2018.04.004
Ali, A. (2019) ‘Forest stand structure and functioning: Current knowledge and future challenges’, Ecological Indicators, 98, pp. 665–677. Available at: https://doi.org/10.1016/j.ecolind.2018.11.017
Ampoorter, E., Barbaro, L., Jactel, H., Baeten, L., Boberg, J., Carnol, M., Castagneyrol, B., Charbonnier, Y., Dawud, S.M., Deconchat, M., Smedt, P. De, Wandeler, H. De, Guyot, V., Hättenschwiler, S., Joly, F., Koricheva, J., Milligan, H., Muys, B., Nguyen, D., Ratcliffe, S., Raulund-Rasmussen, K., Scherer-Lorenzen, M., van der Plas, F., Keer, J. Van, Verheyen, K., Vesterdal, L. and Allan, E. (2020) ‘Tree diversity is key for promoting the diversity and abundance of forest-associated taxa in Europe’, Oikos, 129(2), pp. 133–146. Available at: https://doi.org/10.1111/oik.06290
Anderson, R. (2020) Peatlands, forestry and climate change: what role can forest-to-bog restoration play? Climate Change Factsheet. Forest Research.
Andringa, J.I., Zuo, J., Berg, M.P., Klein, R., van’t Veer, J., de Geus, R., de Beaumont, M., Goudzwaard, L., van Hal, J., Broekman, R., van Logtestijn, R.S.P., Li, Y., Fujii, S., Lammers, M., Hefting, M.M., Sass-Klaassen, U. and Cornelissen, J.H.C. (2019) ‘Combining tree species and decay stages to increase invertebrate diversity in dead wood’, Forest Ecology and Management, 441, pp. 80–88. Available at: https://doi.org/10.1016/j.foreco.2019.03.029
Bellamy, C., Barsoum, N., Cottrell, J. and Watts, K. (2018) ‘Encouraging biodiversity at multiple scales in support of resilient woodlands’, Forestry Commission Research Note, FCRN033.
Bennett, A.F., Radford, J.Q. and Haslem, A. (2006) “Properties of land mosaics: Implications for nature conservation in agricultural environments,” Biological Conservation, 133(2), pp. 250–264. Available at: https://doi.org/10.1016/j.biocon.2006.06.008
Binner, A., Smith, G., Bateman, I., Day, B., Agarwala, M. and Harwood, A. (2017) Valuing the social and environmental contribution of woodlands and trees in England, Scotland and Wales. Forestry Commission Research Report FCRP027.
Bowler, D.E., Cunningham, C.A., Beale, C.M., Emberson, L., Hill, J.K., Hunt, M., Maskell, L., Outhwaite, C.L., White, P.C.L. and Pocock, M.J.O. (2023) ‘Idiosyncratic trends of woodland invertebrate biodiversity in Britain over 45 years’, Insect Conservation and Diversity, 16(6), pp. 776–789. Available at: https://doi.org/10.1111/icad.12685
Broome, A., Inchboard, L.L., Perks, M., Clarke, T.-K., Park, K.J. and Thompson, R. (2021) ‘Can epiphytic lichens of remnant
Atlantic oakwood trees in a planted ancient woodland site survive early stages of woodland restoration?’, Annals of Forest Science, 78(3), p. 58. Available at: https://doi.org/10.1007/s13595-021-01069-w
Broome, A., Stokes, V., Mitchell, R. and Ray, D. (2021) Ecological implications of oak decline in Great Britain. Forest Research, Research Note FRRN040.
Broughton, R.K., Bullock, J.M., George, C., Gerard, F., Maziarz, M., Payne, W.E., Scholefield, P.A., Wade, D. and Pywell, R.F. (2022) ‘Slow development of woodland vegetation and bird communities during 33 years of passive rewilding in open farmland’, PLOS ONE, 17(11), p. e0277545. Available at: https://doi.org/10.1371/journal.pone.0277545
Brown, N.D., Curtis, T. and Adams, E.C. (2015) ‘Effects of clear-felling versus gradual removal of conifer trees on the survival of understorey plants during the restoration of ancient woodlands’, Forest Ecology and Management, 348, pp. 15–22. Available at: https://doi.org/10.1016/j.foreco.2015.03.030
Buechel, M., Slater, L. and Dadson, S. (2022) “Hydrological impact of widespread afforestation in Great Britain using a large ensemble of modelled scenarios,” Communications Earth & Environment, 3(1), p. 6. Available at: https://doi.org/10.1038/s43247-021-00334-0
Burton, V., Moseley, D., Brown, C., Metzger, M.J. and Bellamy, P. (2018) ‘Reviewing the evidence base for the effects of woodland expansion on biodiversity and ecosystem services in the United Kingdom’, Forest Ecology and Management, 430, pp. 366–379. Available at: https://doi.org/10.1016/j.foreco.2018.08.003
Calder, I.R., Harrison, J., Nisbet, T.R. and Smithers, R.J. (2008) Woodland actions for biodiversity and their role in water management. Woodland Trust.
del Campo, A.D., Otsuki, K., Serengil, Y., Blanco, J.A., Yousefpour, R. and Wei, X. (2022) ‘A global synthesis on the effects of thinning on hydrological processes: Implications for forest management’, Forest Ecology and Management, 519(9), p. 120324. Available at: https://doi.org/10.1016/j.foreco.2022.120324.
Cheng, C., Zhang, F., Shi, J. and Kung, H.-T. (2022) ‘What is the relationship between land use and surface water quality? A review and prospects from remote sensing perspective’, Environmental Science and Pollution Research, 29(38), pp. 56887–56907. Available at: https://doi.org/10.1007/s11356-022-21348-x
Cooper, M.M.D., Patil, S.D., Nisbet, T.R., Thomas, H., Smith, A.R. and McDonald, M.A. (2021) ‘Role of forested land for natural flood management in the UK: A review’, WIREs Water, 8(5). Available at: https://doi.org/10.1002/wat2.1541
Cordingley, J.E., Newton, A.C., Rose, R.J., Clarke, R.T., and Bullock, J.M. (2016) ‘Can landscape-scale approaches to conservation management resolve biodiversity–ecosystem service trade-offs?’ Journal of Applied Ecology, 53(1), 96–105. https://doi.org/10.1111/1365-2664.12545
Craven, D., Filotas, E., Angers, V.A. and Messier, C. (2016) “Evaluating resilience of tree communities in fragmented landscapes: linking functional response diversity with landscape connectivity,” Diversity and Distributions, 22(5), pp. 505–518. Available at: https://doi.org/10.1111/ddi.12423
Crick, H.Q.P., Crosher, I.E., Mainstone, C.P., Taylor, S.D., Wharton, A., Langford, P., Larwood, J., Lusardi, J., Appleton, D., Brotherton, P.N.M., Duffield, S.J. and Macgregor, N.A. (2020) Nature Networks Evidence Handbook. Natural England Research Report NERR081. Natural England, York.
Dixon, S.J., Sear, D.A., Odoni, N.A., Sykes, T. and Lane, S.N. (2016) ‘The effects of river restoration on catchment scale flood risk and flood hydrology’, Earth Surface Processes and Landforms, 41(7), pp. 997–1008. Available at: https://doi.org/10.1002/esp.3919
Environment Act 2021, c. 30.
Environmental Targets (Biodiversity) (England) Regulations 2023. (2023). https://www.legislation.gov.uk/uksi/2023/90/ regulation/3/made
Field, E, Hector, A., Barsoum, N. and Koricheva, J. (2025) ‘Tree diversity reduces pathogen damage in temperate forests: A systematic review and meta-analysis’, Forest Ecology and Management, 578(15), p. 122398. Available at: https://doi.org/10.1016/j.foreco.2024.122398
Flowerdew, J.R. and Ellwood, S.A. (2001) ‘Impacts of woodland deer on small mammal ecology’, Forestry: An International Journal of Forest Research, 74(3), pp. 277–287. Available at: https://doi.org/10.1093/forestry/74.3.277
Forestry Commission (2012) Managing forests as red squirrel strongholds. Forestry Commission Practice Note FCPN102.
Forster, E.J., Healey, J.R., Dymond, C. and Styles, D. (2021) ‘Commercial afforestation can deliver effective climate change mitigation under multiple decarbonisation pathways’, Nature Communications, 12(1), p. 3831. Available at: https://doi.org/10.1038/s41467-021-24084-x
Frederickson-Matika, D. and Riddell, C. (2021) Climate change and tree diseases: How are root pathogens likely to be influenced by climate change? Climate Change Factsheet. Forest Research.
Fuentes-Montemayor, E., Ferryman, M., Watts, K., Macgregor, N.A., Hambly, N., Brennan, S., Coxon, R., Langridge, H. and Park, K.J. (2020) ‘Small mammal responses to long-term large-scale woodland creation: the influence of local and landscape-level attributes’, Ecological Applications, 30(2). Available at: https://doi.org/10.1002/eap.2028
Fuentes-Montemayor, E., Goulson, D., Cavin, L., Wallace, J.M. and Park, K.J. (2013) ‘Fragmented woodlands in agricultural landscapes: The influence of woodland character and landscape context on bats and their insect prey’, Agriculture, Ecosystems & Environment, 172(6), pp. 6–15. Available at: https://doi.org/10.1016/j.agee.2013.03.019
Fuentes-Montemayor, E., Park, K.J., Cordts, K. and Watts, K. (2022) ‘The long-term development of temperate woodland creation sites: from tree saplings to mature woodlands’, Forestry: An International Journal of Forest Research, 95(1), pp. 28–37. Available at: https://doi.org/10.1093/forestry/cpab027
Fuller, L., Fuentes-Montemayor, E., Watts, K., Macgregor, N.A., Bitenc, K. and Park, K.J. (2018) ‘Local-scale attributes determine the suitability of woodland creation sites for Diptera’, Journal of Applied Ecology, 55(3), pp. 1173–1184. Available at: https://doi.org/10.1111/1365-2664.13035
Fuller, R.J., Smith, K.W., Grice, P. V., Currie, F.A. and Quine, C.P. (2007) ‘Habitat change and woodland birds in Britain: implications for management and future research’, Ibis, 149(s2), pp. 261–268. Available at: https://doi.org/10.1111/ j.1474-919X.2007.00775.x
Gill, R.M.A. and Fuller, R.J. (2007) ‘The effects of deer browsing on woodland structure and songbirds in lowland Britain’, Ibis, 149(s2), pp. 119–127. Available at: https://doi.org/10.1111/j.1474-919X.2007.00731.x
Harvey, G.L. and Henshaw, A.J. (2023) ‘Rewilding and the water cycle’, WIREs Water, 10(6). Available at: https://doi.org/10.1002/wat2.1686
Hughes, S., Kunin, W., Watts, K. and Ziv, G. (2023) ‘New woodlands created adjacent to existing woodlands grow faster, taller and have higher structural diversity than isolated counterparts’, Restoration Ecology, 31(4). Available at: https://doi.org/10.1111/rec.13889
Humphrey, J., Watts, K., Fuentes-Montemayor, E., Macgregor, and Park, K. (2013) The evidence base for ecological networks: lessons from studies of woodland fragmentation and creation. Forest Research Report.
Humphrey, J.W., Watts, K., Fuentes-Montemayor, E., Macgregor, N.A., Peace, A.J. and Park, K.J. (2015) ‘What can studies of woodland fragmentation and creation tell us about ecological networks? A literature review and synthesis’, Landscape Ecology, 30(1), pp. 21–50. Available at: https://doi.org/10.1007/s10980-014-0107-y
Inward, D. (2023) Climate change and insect pests of trees: Will the impact of damaging forest insects increase? Climate Change Factsheet. Forest Research.
IPBES (2018) IPBES Guide on the production of assessments. Secretariat of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services, Bonn, Germany.
Iremonger, S., Gittings, T., Smith, G.F., Wilson, M., Oxbrough, A., Coote, L., Pithon, J., O’Sullivan, A., Neville, P., Mitchel, F.J.G., O’Donnell, V., Kelly, T. and Dowding, P. (2006) Investigation of experimental methods to enhance biodiversity in plantation forests. BIOFOREST Project 3.1.3 Final Report.
Iversen, S.V., van der Velden, N., Convery, I., Mansfield, L., Kjeldsen, C., Thorsøe, M.H., and Holt, C.D.S (2023) ‘Impacts of woodland planting on nature-based recreational tourism in upland England – A case study’ Landscape and Urban Planning. 230, 104587. Available at: https://doi.org/10.1016/j.landurbplan.2022.104587
Jactel, H., Bauhus, J., Boberg, J., Bonal, D., Castagneyrol, B., Gardiner, B., Gonzalez-Olabarria, J.R., Koricheva, J., Meurisse, and Brockerhoff, E.G. (2017) ‘Tree Diversity Drives Forest Stand Resistance to Natural Disturbances’, Current Forestry Reports, 3(3), pp. 223–243. Available at: https://doi.org/10.1007/s40725-017-0064-1
James, J., Page-Dumroese, D., Busse, M., Palik, B., Zhang, J., Eaton, B., Slesak, R., Tirocke, J. and Kwon, H. (2021) ‘Effects of forest harvesting and biomass removal on soil carbon and nitrogen: Two complementary meta-analyses’, Forest Ecology and Management, 485, p. 118935. Available at: https://doi.org/10.1016/j.foreco.2021.118935.
Kirby, K.J. (2001) ‘The impact of deer on the ground flora of British broadleaved woodland’, Forestry: An International Journal of Forest Research, 74(3), pp. 219–229. Available at: https://doi.org/10.1093/forestry/74.3.219
Kirby, K.J., Buckley, G.P. and Mills, J. (2017) ‘Biodiversity implications of coppice decline, transformations to high forest and coppice restoration in British woodland’, Folia Geobotanica, 52(1), pp. 5–13. Available at: https://doi.org/10.1007/s12224-016-9252-1
Kirby, K.J., Goldberg, E.A. and Orchard, N. (2017) ‘Long-term changes in the flora of oak forests and of oak:spruce mixtures following removal of conifers’, Forestry: An International Journal of Forest Research, 90(1), pp. 136–147. Available at: https://doi.org/10.1093/forestry/cpw049
Körner, C. (2017) ‘A matter of tree longevity’, Science, 355(6321), pp. 130–131. Available at: https://doi.org/10.1126/science.aal2449
Lassauce, A., Paillet, Y., Jactel, H. and Bouget, C. (2011) ‘Deadwood as a surrogate for forest biodiversity: Meta-analysis of correlations between deadwood volume and species richness of saproxylic organisms’, Ecological Indicators, 11(5), pp. 1027–1039. Available at: https://doi.org/10.1016/j.ecolind.2011.02.004
Lawton, J.H., Brotherton, P.N.M., Brown, V.K., Elphick, C., Fitter, A.H., Forshaw, J., Haddow, R.W., Hilborne, S., Leafe, R.N., Mace, G.M., Southgate, M.P.,Sutherland, W.J., Tew, T.E., Varley, J. and Wynne, G.R. (2010) Making Space for Nature: a review of England’s wildlife sites and ecological network. Report to Defra.
Manchester, S.J. and Bullock, J.M. (2000) ‘The impacts of non-native species on UK biodiversity and the effectiveness of control’, Journal of Applied Ecology, 37(5), pp. 845–864. Available at: https://doi.org/10.1046/j.1365-2664.2000.00538.x
Marzano, M and Dandy, N (2012) ‘Recreational use of forests and disturbance of wildlife – a literature review.’ Forestry Commission Research Report. Forestry Commission, Edinburgh. Available at: https://cdn.forestresearch.gov.uk/2012/07/fcrp020.pdf
Matthews, R.W., Henshall, P.A., Beauchamp, K., Gruffudd, H., Hogan, G.P., Mackie, E.D., Sayce, M. and Morison, J.I.L. (2022) Quantifying the sustainable forestry carbon cycle: Summary Report. Forest Research, Farnham.
Mayer, M., Prescott, C.E., Abaker, W.E.A., Augusto, L., Cécillon, L., Ferreira, G.W.D., James, J., Jandl, R., Katzensteiner, K., Laclau, J.-P., Laganière, J., Nouvellon, Y., Paré, D., Stanturf, J.A., Vanguelova, E.I. and Vesterdal, L. (2020) ‘Tamm Review: Influence of forest management activities on soil organic carbon stocks: A knowledge synthesis’, Forest Ecology and Management, 466, p. 118127. Available at: https://doi.org/10.1016/j.foreco.2020.118127
Meeussen, C., Govaert, S., Vanneste, T., Haesen, S., Van Meerbeek, K., Bollmann, K., Brunet, J., Calders, K., Cousins, S.A.O., Diekmann, M., Graae, B.J., Iacopetti, G., Lenoir, J., Orczewska, A., Ponette, Q., Plue, J., Selvi, F., Spicher, F., Sørensen, M.V., Verbeeck, H., Vermeir, P., Verheyen, K., Vangansbeke, P. and De Frenne, P. (2021) ‘Drivers of carbon stocks in forest edges across Europe’, Science of The Total Environment, 759, p. 143497. Available at: https://doi.org/10.1016/j.scitotenv.2020.143497
Merckx, T. (2015) ‘Rewilding: Pitfalls and Opportunities for Moths and Butterflies’, in Rewilding European Landscapes. Cham: Springer International Publishing, pp. 107–125. Available at: https://doi.org/10.1007/978-3-319-12039-3_6
Messier, C., Bauhus, J., Sousa-Silva, R., Auge, H., Baeten, L., Barsoum, N., Bruelheide, H., Caldwell, B., Cavender-Bares, J., Dhiedt, E., Eisenhauer, N., Ganade, G., Gravel, D., Guillemot, J., Hall, J.S., Hector, A., Hérault, B., Jactel, H., Koricheva, J., Kreft, H., Mereu, S., Muys, B., Nock, C.A., Paquette, A., Parker, J.D., Perring, M.P., Ponette, Q., Potvin, C., Reich, P.B., Scherer-Lorenzen, M., Schnabel, F., Verheyen, K., Weih, M., Wollni, and Zemp, D.C. (2022) ‘For the sake of resilience and multifunctionality, let’s diversify planted forests!’, Conservation Letters, 15(1). Available at: https://doi.org/10.1111/conl.12829
Milner, A.M., Baird, A.J., Davidson, S.J., Lines, E.R., Abrahams, D., Ahiable, C.A.E., Barsoum, N., Bryant, M., Dear, E., Diack, I., Duley, E., Noach, A., Roland, T.P. and Smedley, D. (2024) ‘The forgotten forests: Incorporating temperate peat-forming wet woodlands as nature-based solutions into policy and practice’, Ecological Solutions and Evidence, 5(2). Available at: https://doi.org/10.1002/2688-8319.12346
Mina, M., Messier, C., Duveneck, M.J., Fortin, M. and Aquilué, N (2022) “Managing for the unexpected: Building resilient forest landscapes to cope with global change,” Global Change Biology, 28(14), pp. 4323–4341. Available at: https:// org/10.1111/gcb.16197
Mitchell, R.J., Beaton, J.K., Bellamy, P.E., Broome, A., Chetcuti, J., Eaton, S., Ellis, C.J., Gimona, A., Harmer, R., Hester, A.J., Hewison, R.L., Hodgetts, N.G., Iason, G.R., Kerr, G., Littlewood, N.A., Newey, S., Potts, J.M., Pozsgai, G., Ray, D., Sim, D.A., Stockan, J.A., Taylor, A.F.S. and Woodward, S. (2014) ‘Ash dieback in the UK: A review of the ecological and conservation implications and potential management options’, Biological Conservation, 175, pp. 95–109. Available at: https://doi.org/10.1016/j.biocon.2014.04.019
Mitchell, R.J., Hewison, R.L., Haghi, R.K., Robertson, A.H.J., Main, A.M. and Owen, I.J. (2021) ‘Functional and ecosystem service differences between tree species: implications for tree species replacement’, Trees, 35(1), pp. 307–317. Available at: https://doi.org/10.1007/s00468-020-02035-1
Morison, J., Matthews, R., Miller, G., Perks, M., Randle, T., Vanguelova, E., White, M. and Yamulki, S. (2012) Understanding the carbon and greenhouse gas balance of forests in Britain. Forestry Commission Research Report. Forestry Commission, Edinburgh.
Morreale, L.L., Thompson, J.R., Tang, X., Reinmann, A.B. and Hutyra, L.R. (2021) ‘Elevated growth and biomass along temperate forest edges’, Nature Communications, 12(1), 7181. Available at: https://doi.org/10.1038/s41467-021-27373-7
National Forest Inventory (2020) ‘NFI woodland ecological condition in Great Britain: Statistics.’ Forest Research.
Nisbet, T. (2020) Climate change, flooding and forests: how can forestry help reduce flooding? Climate Change Factsheet. Forest Research.
Nisbet, T., Silgram, M., Shah, N., Morrow, K. and Broadmeadow, S, (2011) Woodland for Water: Woodland measures for meeting Water Framework Directive objectives. Forest Research Monograph 4.
Nisbet, T.R. and Evans, C.D. (2014) ‘Forestry and surface water acidification’, Forestry Commission Research Note, FCRN016
Office for National Statistics (ONS) (2024) ‘Woodland natural capital accounts, UK: 2024.’ Available at: https://www.ons.gov.uk/economy/environmentalaccounts/bulletins/ woodlandnaturalcapitalaccountsuk/2024
Oliver, T.H., Heard, M.S., Isaac, N.J.B., Roy, D.B., Procter, D., Eigenbrod, F., Freckleton, R., Hector, A., Orme, C.D.L., Petchey, O.L., Proença, V., Raffaelli, D., Suttle, K.B., Mace, G.M., Martín-López, B., Woodcock, B.A. and Bullock, J.M. (2015) “Biodiversity and Resilience of Ecosystem Functions,” Trends in Ecology & Evolution, 30(11), pp. 673–684. Available at: https://doi.org/10.1016/j.tree.2015.08.009
Patacca, M., Lindner, M., Lucas-Borja, M.E., Cordonnier, T., Fidej, G., Gardiner, B., Hauf, Y., Jasinevičius, G., Labonne, S., Linkevičius, E., Mahnken, M., Milanovic, S., Nabuurs, G., Nagel, T.A., Nikinmaa, L., Panyatov, M., Bercak, R., Seidl, R., Ostrogović Sever, M.Z., Socha, J., Thom, D., Vuletic,
D., Zudin, S. and Schelhaas, M. (2023) ‘Significant increase in natural disturbance impacts on European forests since 1950’, Global Change Biology, 29(5), pp. 1359–1376. Available at: https://doi.org/10.1111/gcb.16531
Powe, N.A., and Willis, K.G. (2004) ‘Mortality and morbidity benefits of air pollution (SO2 and PM10) absorption attributable to woodland in Britain.’ Journal of Environmental Management. 70(2), 119–128. https://doi.org/10.1016/j.jenvman.2003.11.003
Prevedello, J.A. and Vieira, M.V. (2010) ‘Does the type of matrix matter? A quantitative review of the evidence’, Biodiversity and Conservation, 19(5), pp. 1205–1223. Available at: https://doi.org/10.1007/s10531-009-9750-z
Quine, C.P., Fuller, R.J., Smith, K.W. and Grice, P. V. (2007) ‘Stand management: a threat or opportunity for birds in British woodland?’, Ibis, 149(s2), pp. 161–174. Available at: https://doi.org/10.1111/j.1474-919X.2007.00742.x
Ramirez, J.I., Jansen, P.A. and Poorter, L. (2018) ‘Effects of wild ungulates on the regeneration, structure and functioning of temperate forests: A semi-quantitative review’, Forest Ecology and Management, 424, pp. 406–419. Available at: https://doi.org/10.1016/j.foreco.2018.05.016
Ray, D., Morison, J. and Broadmeadow, M. (2010) ‘Climate change: impacts and adaptation in England’s woodlands’, Forestry Commission Research Note, FCRN201.
Riva, F. and Fahrig, L. (2022) ‘The disproportionately high value of small patches for biodiversity conservation’, Conservation Letters, 15(3). Available at: https://doi.org/10.1111/conl.12881
Roberts, M., Gilligan, C.A., Kleczkowski, A., Hanley, N., Whalley, A.E. and Healey, J.R. (2020) ‘The Effect of Forest Management Options on Forest Resilience to Pathogens’, Frontiers in Forests and Global Change, 3. Available at: https://doi.org/10.3389/ffgc.2020.00007
Scheidegger, C. and Werth, S. (2009) ‘Conservation strategies for lichens: insights from population biology’, Fungal Biology Reviews, 23(3), pp. 55–66. Available at: https://doi.org/10.1016/j.fbr.2009.10.003
Seddon, N., Smith, A., Smith, P., Key, I., Chausson, A., Girardin, C., House, J., Srivastava, S. and Turner, B. (2021) ‘Getting the message right on nature-based solutions to climate change’, Global Change Biology, 27(8), pp. 1518–1546. Available at: https://doi.org/10.1111/gcb.15513
Sing, L., Metzger, M.J., Paterson, J.S. and Ray, D. (2018) “A review of the effects of forest management intensity on ecosystem services for northern European temperate forests with a focus on the UK,” Forestry: An International Journal of Forest Research, 91(2), pp. 151–164. Available at: https://doi.org/10.1093/forestry/cpx042
Stewart, A.J.A. (2001) ‘The impact of deer on lowland woodland invertebrates: a review of the evidence and priorities for future research’, Forestry: An International Journal of Forest Research, 74(3), pp. 259–270. Available at: https://doi.org/10.1093/forestry/74.3.259
Stewart, P.S., Hill, R.A., Stephens, P.A., Whittingham, M.J. and Dawson, W. (2021) ‘Impacts of invasive plants on animal behaviour’, Ecology Letters, 24(4), pp. 891–907. Available at: https://doi.org/10.1111/ele.13687
Sweeney, O.F.McD., Wilson, M.W., Irwin, S., Kelly, T.C. and O’Halloran, J. (2010) ‘Breeding bird communities of second-rotation plantations at different stages of the forest cycle’, Bird Study, 57(3), pp. 301–314. Available at: https://doi.org/10.1080/00063651003801713
Synes, N. W., Ponchon, A., Palmer, S. C. F., Osborne, P. E., Bocedi, G., Travis, J. M. J., and Watts, K. (2020) ‘Prioritising conservation actions for biodiversity: Lessening the impact from habitat fragmentation and climate change.’ Biological Conservation, 252, 108819. Available at: https://doi.org/10.1016/j.biocon.2020.108819
Tanentzap, A.J. and Coomes, D.A. (2012) ‘Carbon storage in terrestrial ecosystems: do browsing and grazing herbivores matter?’, Biological Reviews, 87(1), pp. 72–94. Available at: https://doi.org/10.1111/j.1469-185X.2011.00185.x
Tomao, A., Antonio Bonet, J., Castaño, C. and de-Miguel, S. (2020) ‘How does forest management affect fungal diversity and community composition? Current knowledge and future perspectives for the conservation of forest fungi’, Forest Ecology and Management, 457, p. 117678. Available at: https://doi.org/10.1016/j.foreco.2019.117678
Trout, R.C., Brooks, S.E., Rudlin, P. and Neil, J. (2012) ‘The effects of restoring a conifer Plantation on an Ancient Woodland Site (PAWS) in the UK on the habitat and local population of the Hazel Dormouse (Muscardinus avellanarius)’, European Journal of Wildlife Research, 58(4), pp. 635–643. Available at: https://doi.org/10.1007/s10344-012-0611-9
Vilà, M., Espinar, J.L., Hejda, M., Hulme, P.E., Jarošík, V., Maron, J.L., Pergl, J., Schaffner, U., Sun, Y. and Pyšek, P. (2011) ‘Ecological impacts of invasive alien plants: a meta-analysis of their effects on species, communities and ecosystems’, Ecology Letters, 14(7), pp. 702–708. Available at: https://doi.org/10.1111/j.1461-0248.2011.01628.x
Waddell, E.H., Fuentes-Montemayor, E., Park, K.J., Carey, P., Guy, M., Macgregor, N.A. and Watts, K. (2024) ‘Larger and structurally complex woodland creation sites provide greater benefits for woodland plants’, Ecological Solutions and Evidence, in press.
Warner, E., Cook-Patton, S.C., Lewis, O.T., Brown, N., Koricheva, J., Eisenhauer, N., Ferlian, O., Gravel, D., Hall, J.S., Jactel, H., Mayoral, C., Meredieu, C., Messier, C., Paquette, A., Parker, W.C., Potvin, C., Reich, P.B. and Hector, A. (2023) ‘Young mixed planted forests store more carbon than monocultures—a meta-analysis’, Frontiers in Forests and Global Change, 6. Available at: https://doi.org/10.3389/ffgc.2023.1226514
Watts, K., Whytock, R.C., Park, K.J., Fuentes-Montemayor, E., Macgregor, N.A., Duffield, S. and McGowan, P.J.K. (2020) ‘Ecological time lags and the journey towards conservation success’, Nature Ecology & Evolution, 4(3), pp. 304–311. Available at: https://doi.org/10.1038/s41559-019-1087-8
Whytock, R.C., Fuentes-Montemayor, E., Watts, K., Barbosa De Andrade, P., Whytock, R.T., French, P., Macgregor, N.A. and Park, K.J. (2018) ‘Bird-community responses to habitat creation in a long-term, large-scale natural experiment’, Conservation Biology, 32(2), pp. 345–354. Available at: https://doi.org/10.1111/cobi.12983
Wilkes, M.A., Bennett, J., Burbi, S., Charlesworth, S., Dehnen-Schmutz, K., Rayns, F., Schmutz, U., Smith, B., Tilzey, M., Trenchard, L., and van de Wiel, M. (2020) ‘Making Way for Trees? Changes in Land-Use, Habitats and Protected Areas in Great Britain under “Global Tree Restoration Potential.”’ Sustainability, 12(14), 5845. Available at: https://doi.org/10.3390/su12145845
Williams, L.J., Paquette, A., Cavender-Bares, J., Messier, C. and Reich, P.B. (2017) ‘Spatial complementarity in tree crowns explains overyielding in species mixtures’, Nature Ecology & Evolution, 1(4), p. 0063. Available at: https://doi.org/10.1038/s41559-016-0063
Wilson, M.W., Pithon, J., Gittings, T., Kelly, T.C., Giller, P.S. and O’Halloran, J. (2006) ‘Effects of growth stage and tree species composition on breeding bird assemblages of plantation forests’, Bird Study, 53(3), pp. 225–236. Available at: https://doi.org/10.1080/00063650609461437
Zeller, L., Förster, A., Keye, C., Meyer, P., Roschak, C. and Ammer, (2023) ‘What does literature tell us about the relationship between forest structural attributes and species richness in temperate forests? – A review’, Ecological Indicators, 153, p. 110383. Available at: https://doi.org/10.1016/j.ecolind.2023.110383

